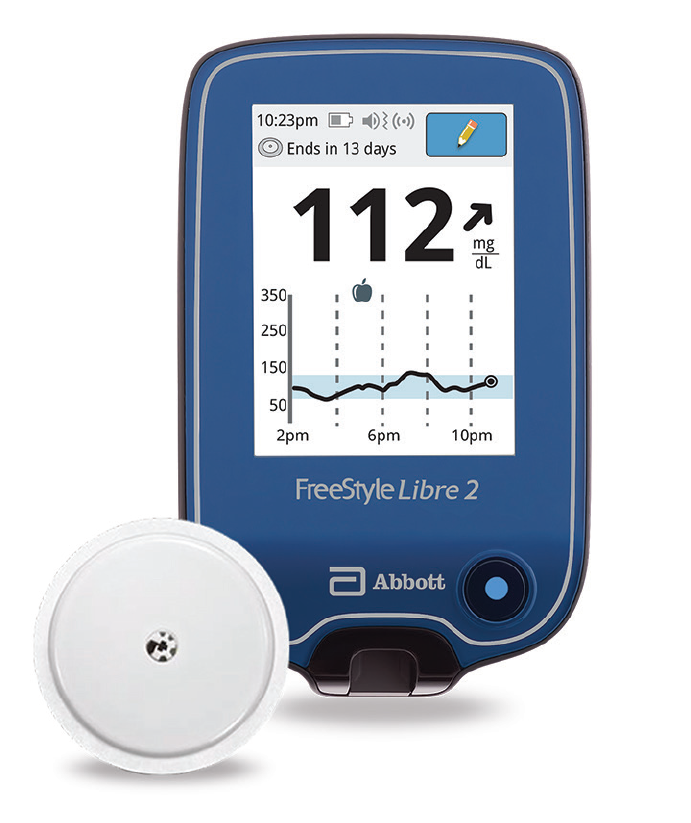
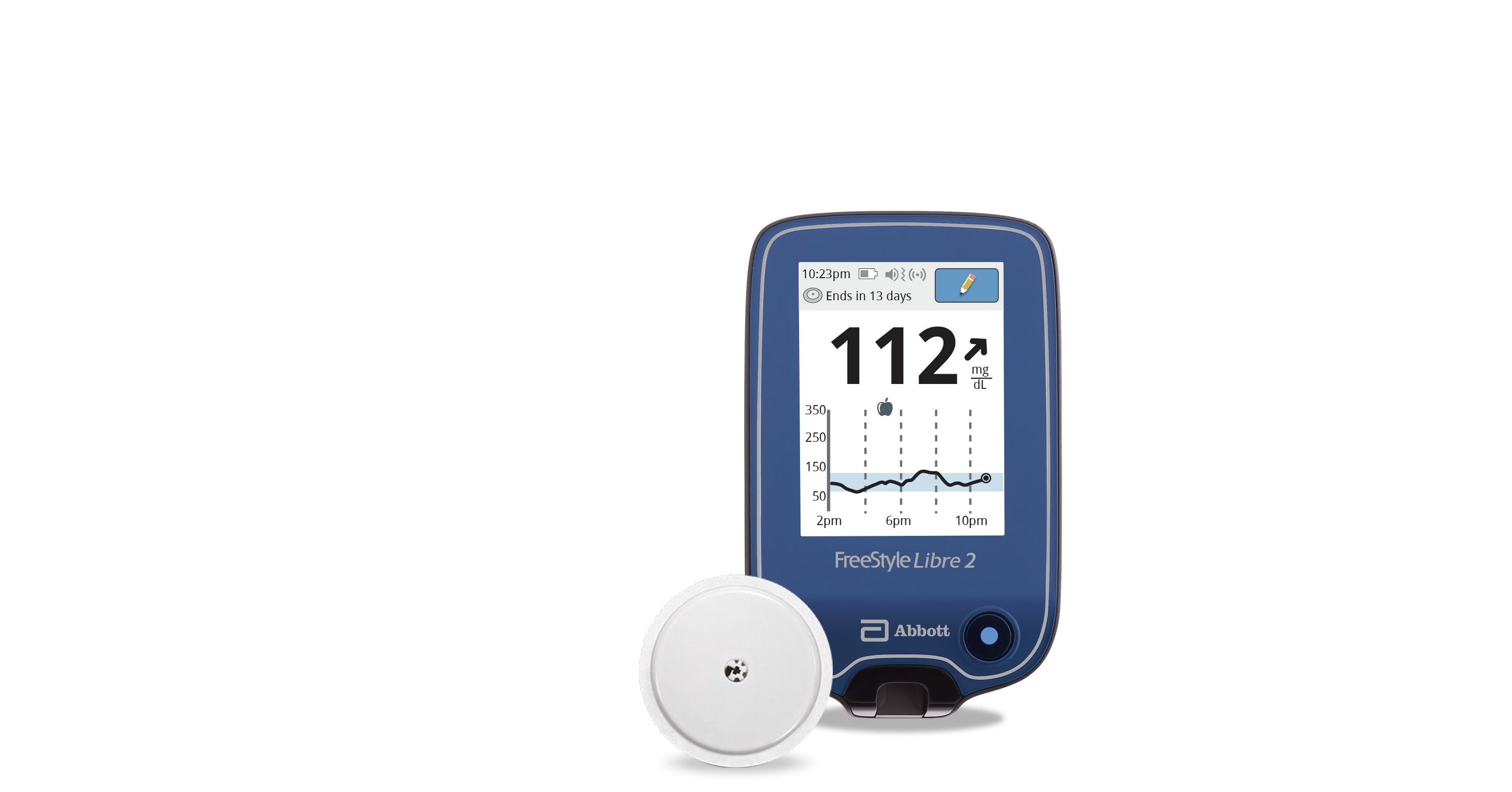
Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards - Jun 15, 2020
Abbott (NYSE: ABT), the worldwide leader in continuous glucose monitoring (CGM), announced today the U.S. Food and Drug Administration (FDA) cleared its next-generation FreeStyle® Libre 2
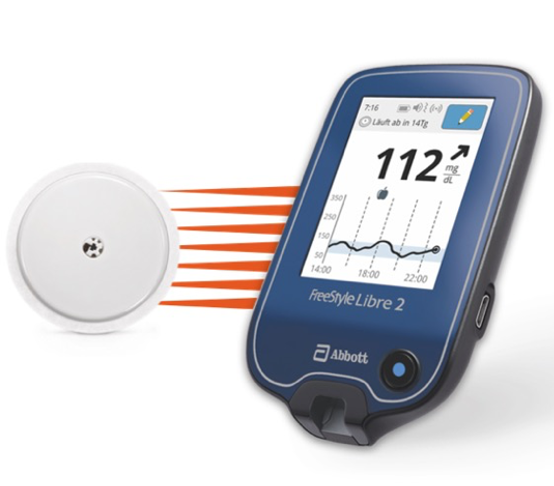
FreeStyle Libre 2 Cleared by FDA as iCGM

Emerging Diabetes Technologies: Continuous Glucose Monitors/Artificial Pancreases

Improving Equitable Access to Continuous Glucose Monitors for Alabama's Children with Type 1 Diabetes: A Quality Improvement Project

Abbott's FreeStyle® Libre 2 iCGM Cleared in U.S. for Adults and Children with Diabetes, Achieving Highest Level of Accuracy and Performance Standards

FreeStyle Libre 2 Continuous Glucose Monitor
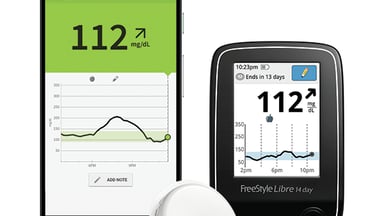
ADA: Real-world data show lower A1c levels with Abbott's FreeStyle Libre CGM in Type 2 diabetes

FreeStyle Libre 3: World's Smallest Sensor

Dexcom G6 CGM System for Personal Use
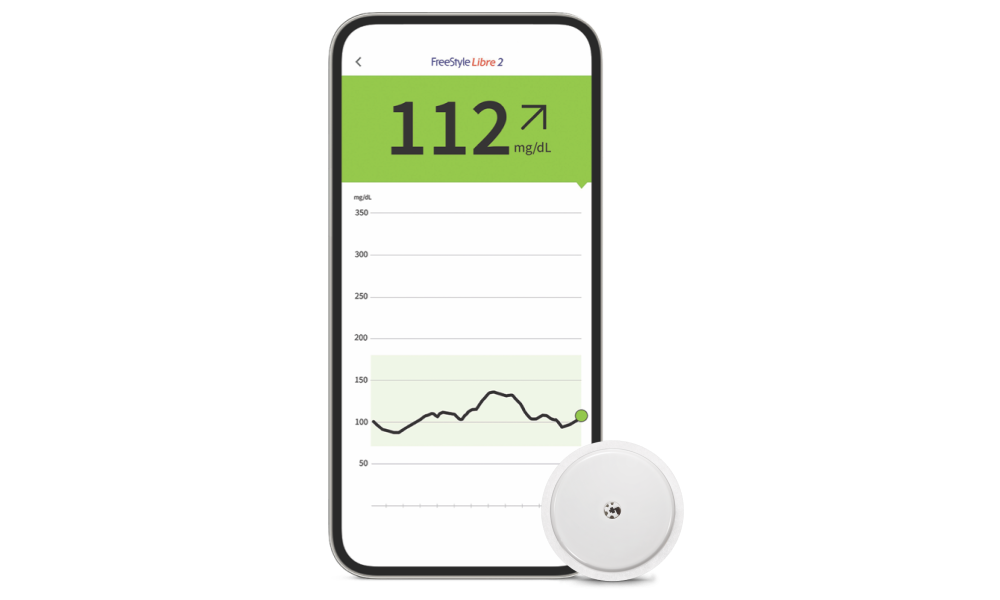
FreeStyle Libre 2 System (CGM)

Diabetes Technology Meeting 2020 - Trisha Shang, Jennifer Y. Zhang, B. Wayne Bequette, Jennifer K. Raymond, Gerard Coté, Jennifer L. Sherr, Jessica Castle, John Pickup, Yarmela Pavlovic, Juan Espinoza, Laurel H. Messer
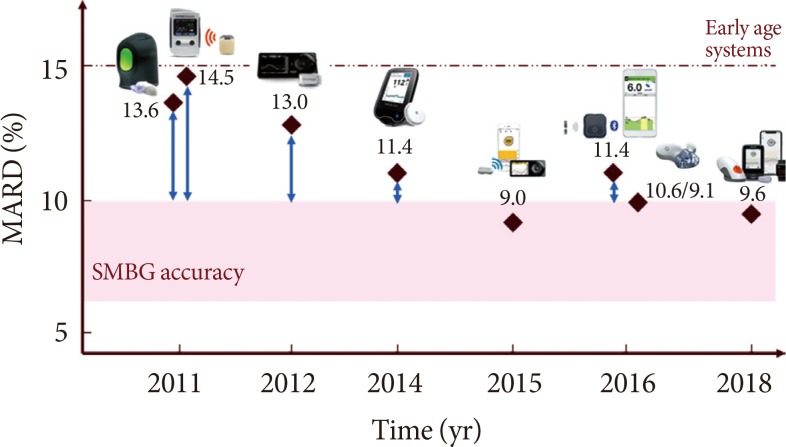
Continuous Glucose Monitoring Sensors for Diabetes Management: A Review of Technologies and Applications

FreeStyle Libre 2 Continuous Glucose Monitor

FreeStyle Libre 2 Reader - Healthcare Home Medical Supply USA

2 Million Patients And Beyond: Abbott's FreeStyle Libre 2 Cleared In The U.S. For Adults And Children With Diabetes
Abbott® Freestyle Libre 2 Sensor - Continuous Glucose Monitoring Device (CGM) - Advanced Diabetes Supply®